Stay updated on Amgen Press Releases
Sign up to get notified when there's something new on the Amgen Press Releases page.
Latest updates to the Amgen Press Releases page
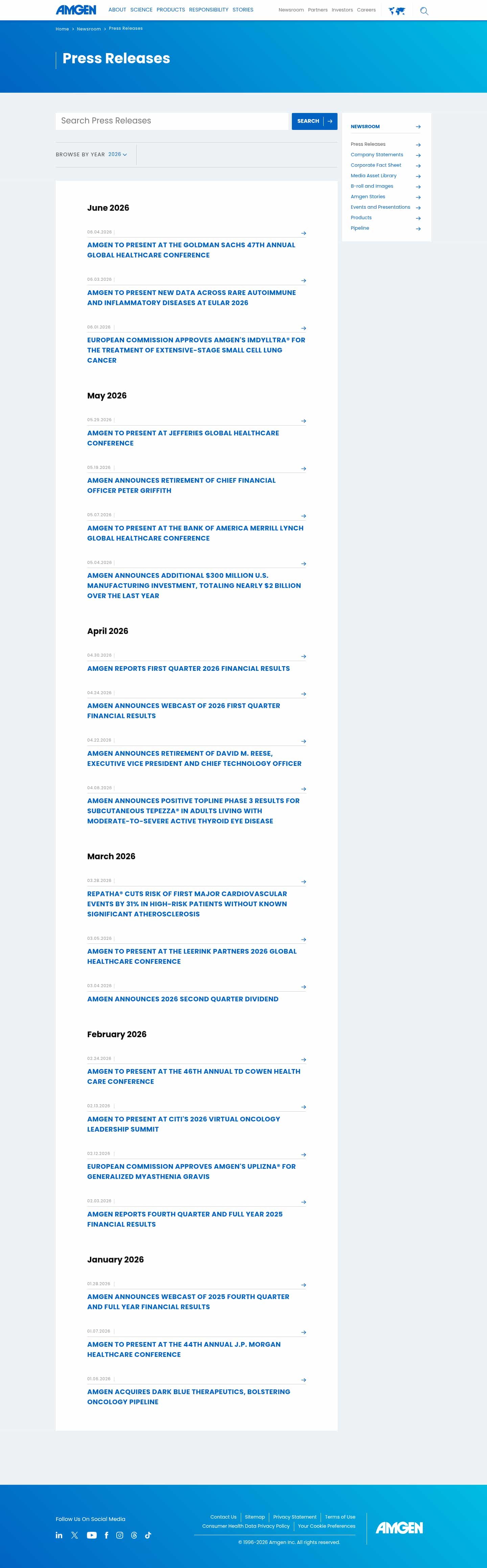
- Check5 days agoChange DetectedFour new press releases have been added to the Newsroom for June 2026: the EU approval of Imdylltra for extensive-stage small cell lung cancer, new data to be presented at EULAR 2026, and invitations to present at the Goldman Sachs 47th Annual Global Healthcare Conference and the Jefferies Global Healthcare Conference.SummaryDifference3%
- Check12 days agoNo Change Detected
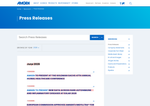
- Check19 days agoChange DetectedA new press release announcing the retirement of CFO Peter Griffith was added to the May 2026 listings, and several header elements and social icons were removed from the page layout.SummaryDifference12%

- Check27 days agoNo Change Detected
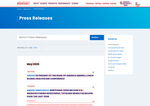
- Check34 days agoChange DetectedThe Newsroom Press Releases page now lists new entries for May and April 2026. It also includes branding and navigation elements (logo, search, social icons) to improve browsing and discovery of releases.SummaryDifference13%
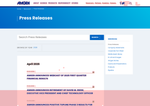
- Check41 days agoChange DetectedNew press release items with dates have been added to Amgen's Newsroom Press Releases, expanding the list of announcements visible to readers. At the same time, several header and social-navigation UI elements were removed, altering the page layout.SummaryDifference21%
- Check48 days agoChange DetectedThe press releases list under the 2026 browse-by-year section has removed multiple dated news items, including an acquisition headline, financial results and webcast announcements, a dividend announcement, regulatory approval for UPLIZNA®, conference presentation notices, and other headline scientific updates.SummaryDifference8%
- Check55 days agoChange DetectedA search control was added to the Press Releases page, allowing users to search press releases by keyword. This enhances navigation and quick access to specific announcements.SummaryDifference0.3%
Stay in the know with updates to Amgen Press Releases
Enter your email address, and we'll notify you when there's something new on the Amgen Press Releases page.