Stay updated on Darolutamide in Hormone Sensitive Prostate Cancer Clinical Trial
Sign up to get notified when there's something new on the Darolutamide in Hormone Sensitive Prostate Cancer Clinical Trial page.
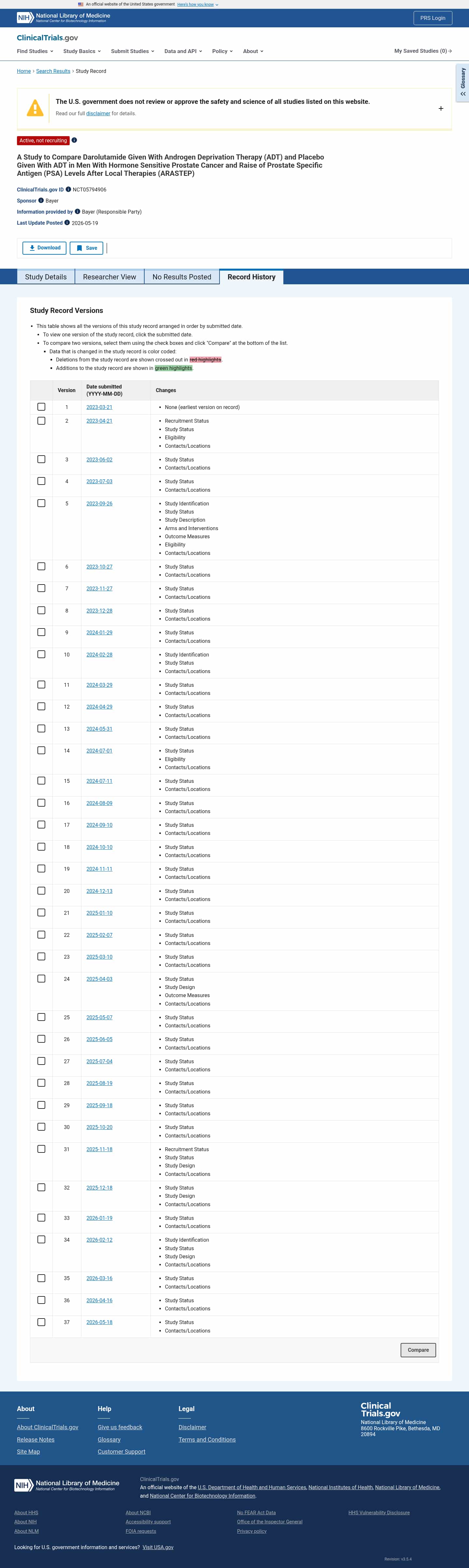
Latest updates to the Darolutamide in Hormone Sensitive Prostate Cancer Clinical Trial page
- Check5 days agoNo Change Detected
- Check12 days agoChange DetectedOn 2026-05-19, Contacts/Locations and Study Status were added to the record history. The version count is shown as 37, and the entry from 2026-04-17 was removed.SummaryDifference0.3%
- Check19 days agoChange DetectedA new revision entry (v3.5.4) was added to the record history, indicating the study record has been updated. The prior revision (v3.5.3) was removed from the current change set.SummaryDifference0.1%
- Check26 days agoNo Change Detected
- Check40 days agoChange DetectedThe displayed record history list reflects a new submitted version dated 2026-04-17 with updates labeled for Contacts/Locations and Study Status, and it also shows an interface revision update to v3.5.3. An earlier revision reference (v3.5.2) is removed from the comparison list.SummaryDifference0.4%
- Check47 days agoChange DetectedAdded Revision: v3.5.2 and removed Revision: v3.5.0 from the history.SummaryDifference0.1%
- Check76 days agoChange DetectedVersion 35, submitted on 2026-03-16, adds updates to Study Status and Contacts/Locations and updates the record revision to v3.5.0.SummaryDifference0.4%
- Check83 days agoChange DetectedFooter revision updated from v3.4.2 to v3.4.3.SummaryDifference0.1%
Stay in the know with updates to Darolutamide in Hormone Sensitive Prostate Cancer Clinical Trial
Enter your email address, and we'll notify you when there's something new on the Darolutamide in Hormone Sensitive Prostate Cancer Clinical Trial page.