Stay updated on Pediatric InO Study in First Relapse ALL Clinical Trial
Sign up to get notified when there's something new on the Pediatric InO Study in First Relapse ALL Clinical Trial page.
Latest updates to the Pediatric InO Study in First Relapse ALL Clinical Trial page
- ChecktodayNo Change Detected
- Check7 days agoNo Change Detected
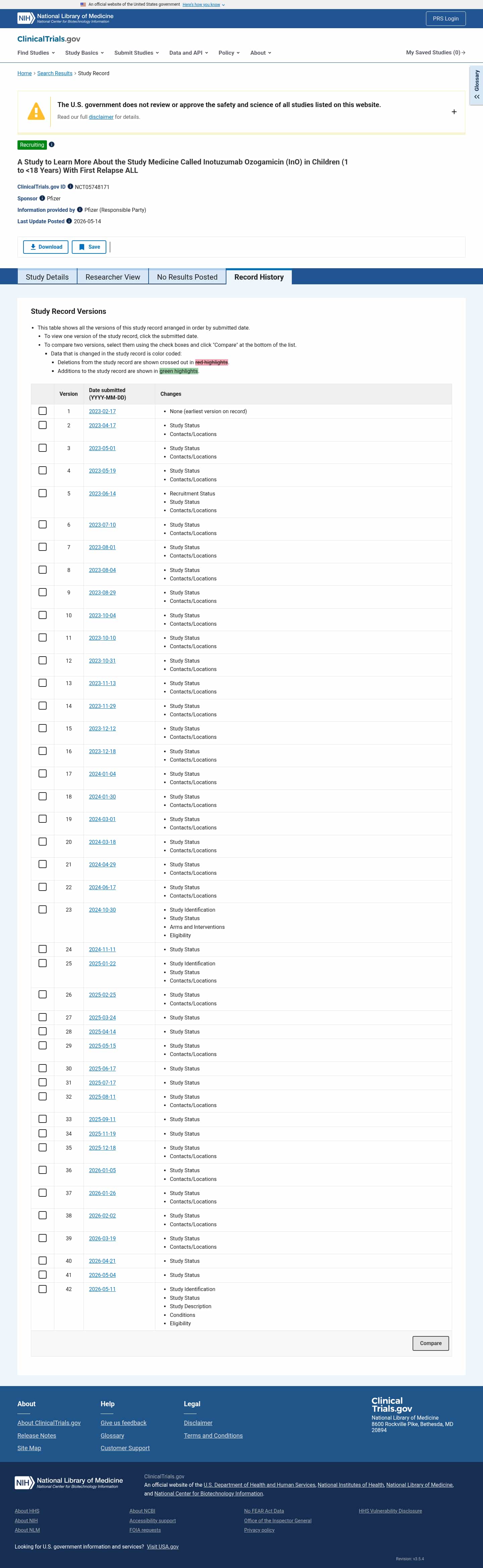
- Check15 days agoChange DetectedA new version 42 was added on 2026-05-11 updating Study Identification and Study Status. On 2026-05-14 a new Revision v3.5.4 was introduced, updating Eligibility, Conditions, and Study Description, while the earlier Revision v3.5.3 was removed on 2026-05-05.SummaryDifference0.6%
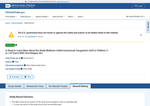
- Check22 days agoChange DetectedA new record version (Version 41) was added on 2026-05-04 with updates to Study Status and Contacts/Locations; a prior entry dated 2026-04-23 was removed from the history.SummaryDifference0.3%
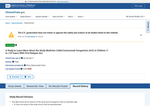
- Check29 days agoChange DetectedRevision: v3.5.3 appears in the Record History, replacing v3.5.2. This reflects a minor site update to the page without altering trial data or results.SummaryDifference0.1%
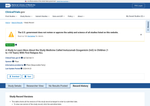
- Check36 days agoChange DetectedThe study record history shows a new version submitted on 2026-04-21/2026-04-23 with changes limited to the study status. No other substantive study information is indicated as added or removed.SummaryDifference0.3%
- Check43 days agoChange DetectedRevision: v3.5.2 is now displayed as the latest revision and Revision: v3.5.0 is removed. This is a minor metadata update to the page's version labeling.SummaryDifference0.1%
- Check65 days agoChange DetectedAdded Contacts/Locations and Study Status entries to the record history; a deletion dated 2026-02-03 was recorded as part of the updates.SummaryDifference0.3%
Stay in the know with updates to Pediatric InO Study in First Relapse ALL Clinical Trial
Enter your email address, and we'll notify you when there's something new on the Pediatric InO Study in First Relapse ALL Clinical Trial page.