Stay updated on Safety and Efficacy of Sparsentan in IgA Nephropathy Clinical Trial
Sign up to get notified when there's something new on the Safety and Efficacy of Sparsentan in IgA Nephropathy Clinical Trial page.
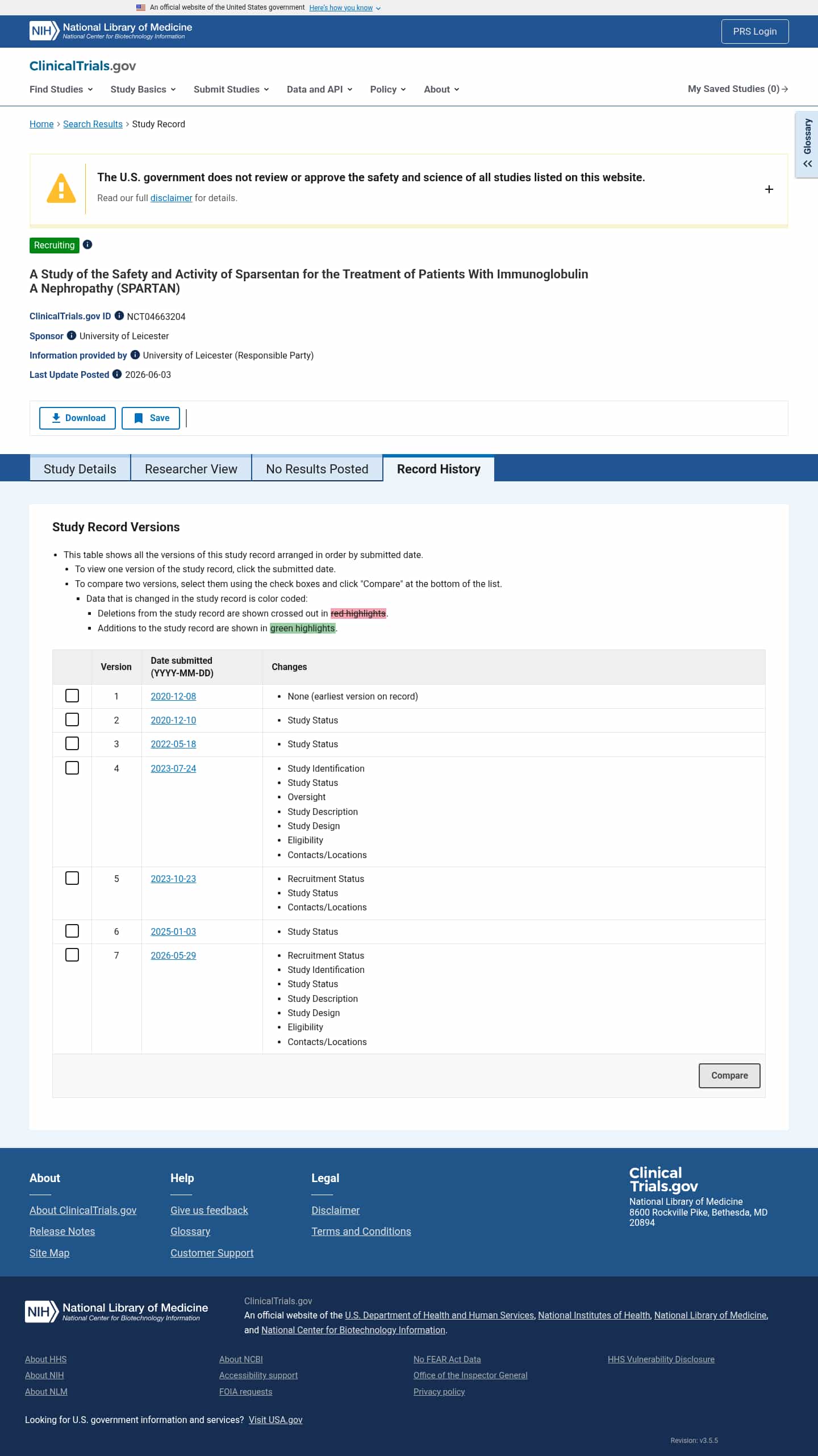
Latest updates to the Safety and Efficacy of Sparsentan in IgA Nephropathy Clinical Trial page
- CheckyesterdayChange DetectedThe study history now marks SPARTAN as Recruiting and updates the page to reflect current enrollment. It also adds key sections such as Contacts/Locations, Eligibility, Study Design, Study Description, Study Status, Study Identification, and Recruitment Status, replacing the older 'Active, not recruiting' label and the previous trial title wording.SummaryDifference1%
- Check8 days agoNo Change Detected
- Check15 days agoNo Change Detected
- Check22 days agoChange DetectedA new revision entry v3.5.4 is added to the record history, updating the study's version history. The previous revision v3.5.3 is removed from the list.SummaryDifference0.1%
- Check43 days agoChange DetectedThe page’s footer updates the ClinicalTrials.gov build/release revision from **v3.5.2** to **v3.5.3**, indicating a web/version update. No study-specific content or record-history details are altered.SummaryDifference0.1%
- Check50 days agoChange DetectedAdded Revision: v3.5.2 to the history. Removed Revision: v3.5.0.SummaryDifference0.1%
- Check79 days agoChange DetectedRevision v3.5.0 was added and revision v3.4.3 was removed from the Record History.SummaryDifference0.1%
- Check86 days agoChange DetectedRecord History now shows Revision: v3.4.3 and removes Revision: v3.4.2.SummaryDifference0.1%

Stay in the know with updates to Safety and Efficacy of Sparsentan in IgA Nephropathy Clinical Trial
Enter your email address, and we'll notify you when there's something new on the Safety and Efficacy of Sparsentan in IgA Nephropathy Clinical Trial page.