Stay updated on Safety of IV ABBV-383 in Multiple Myeloma Clinical Trial
Sign up to get notified when there's something new on the Safety of IV ABBV-383 in Multiple Myeloma Clinical Trial page.
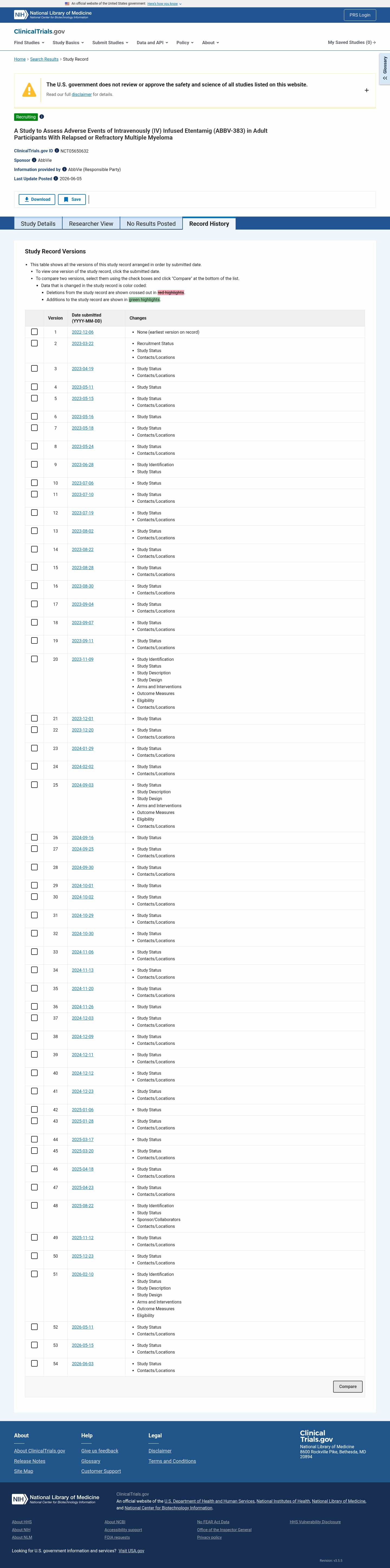
Latest updates to the Safety of IV ABBV-383 in Multiple Myeloma Clinical Trial page
- Check5 days agoChange DetectedA new revision (v3.5.5) on 2026-06-05 adds the Contacts/Locations and Study Status sections to the record, updating the study with current contact details and status information.SummaryDifference0.3%
- Check12 days agoNo Change Detected
- Check19 days agoChange DetectedThe history adds entries on 2026-05-19 for Contacts/Locations and for Study Status, indicating updates to contact information and the trial's current status. An additional item labeled '53' was added on 2026-05-15, and a deletion occurred on 2026-05-13.SummaryDifference0.3%
- Check26 days agoChange DetectedVersion 52 was added on 2026-05-11, updating Study Status and Contacts/Locations for this trial record. A revision entry, v3.5.4, was recorded on 2026-05-13 to reflect those updates.SummaryDifference0.3%
- Check47 days agoChange DetectedThe page’s footer/version identifier updates from “Revision: v3.5.2” to “Revision: v3.5.3,” indicating an internal UI release rather than a change to the recorded clinical trial information.SummaryDifference0.1%
- Check54 days agoChange DetectedAdded Revision: v3.5.2; Deleted Revision: v3.5.0.SummaryDifference0.1%
- Check83 days agoChange DetectedNew revision entry v3.5.0 was added to the record history. Revision v3.4.3 was removed.SummaryDifference0.1%
Stay in the know with updates to Safety of IV ABBV-383 in Multiple Myeloma Clinical Trial
Enter your email address, and we'll notify you when there's something new on the Safety of IV ABBV-383 in Multiple Myeloma Clinical Trial page.