Stay updated on TQ05105 Tablets in Refractory aGVHD Clinical Trial
Sign up to get notified when there's something new on the TQ05105 Tablets in Refractory aGVHD Clinical Trial page.
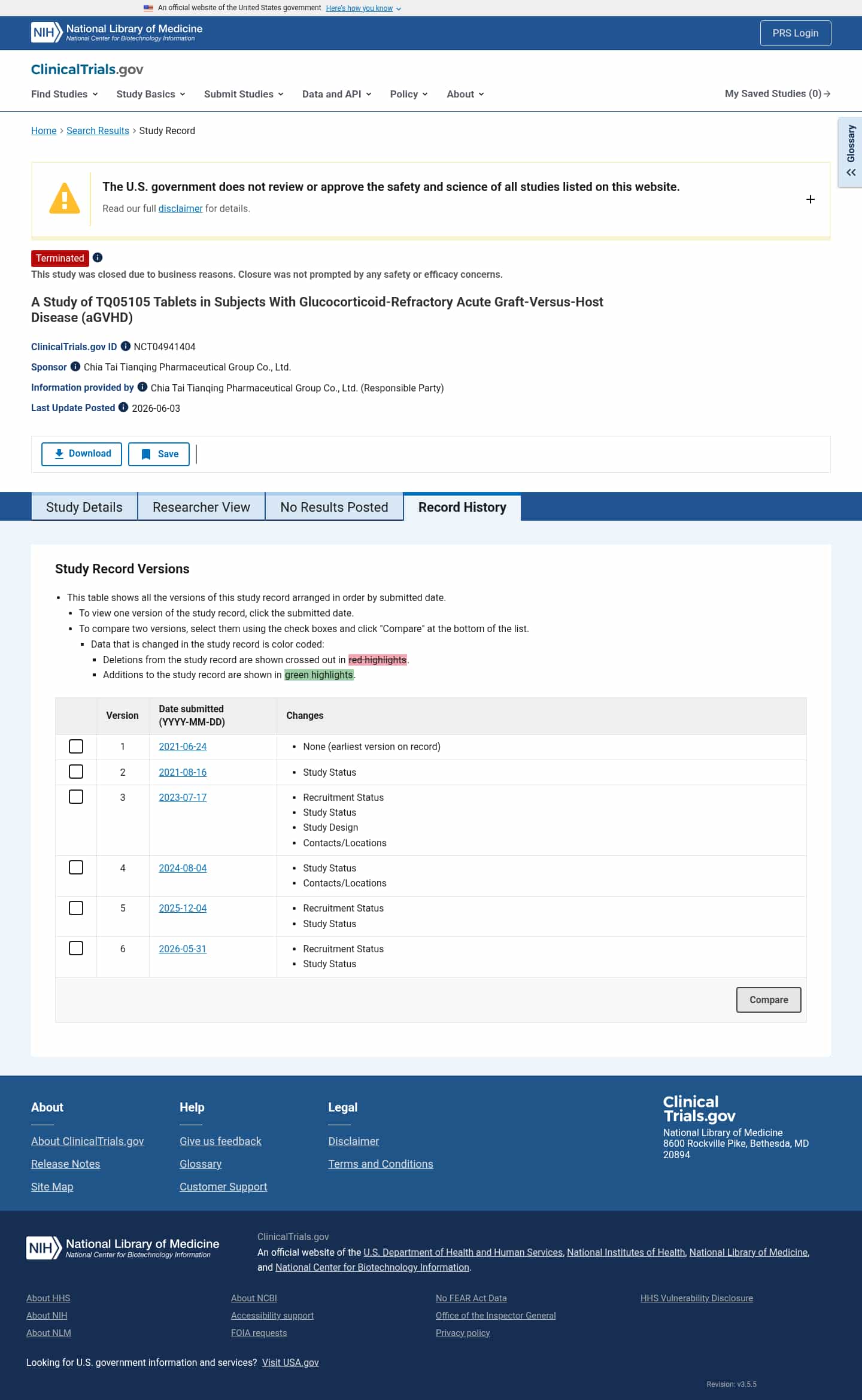
Latest updates to the TQ05105 Tablets in Refractory aGVHD Clinical Trial page
- Check5 days agoChange DetectedThe history adds that the study was closed and terminated for business reasons, with a note that safety and efficacy concerns did not drive the closure. A new revision (v3.5.5) is posted and the previous 'Completed' status was removed.SummaryDifference0.9%
- Check12 days agoNo Change Detected
- Check19 days agoNo Change Detected
- Check26 days agoChange DetectedFooter now shows Revision: v3.5.4 instead of v3.5.3, reflecting a new release version of the record history page. This update appears to be a metadata update rather than a modification to the study information.SummaryDifference0.1%
- Check40 days agoChange DetectedA new revision v3.5.3 was added to the study's record history, indicating an update to the trial record. The previous revision v3.5.2 was removed from the history view.SummaryDifference0.1%
- Check55 days agoChange DetectedRevision v3.5.2 was added to the record history and revision v3.5.0 was removed.SummaryDifference0.1%
- Check83 days agoChange DetectedA new revision entry v3.5.0 was added to the record history, replacing the previous v3.4.3.SummaryDifference0.1%
- Check90 days agoChange DetectedA new latest revision, v3.4.3, is added to the history and the previous revision v3.4.2 is removed.SummaryDifference0.1%
Stay in the know with updates to TQ05105 Tablets in Refractory aGVHD Clinical Trial
Enter your email address, and we'll notify you when there's something new on the TQ05105 Tablets in Refractory aGVHD Clinical Trial page.