Stay updated on Weekly Insulin Icodec with Semaglutide in Type 2 Diabetes Clinical Trial
Sign up to get notified when there's something new on the Weekly Insulin Icodec with Semaglutide in Type 2 Diabetes Clinical Trial page.
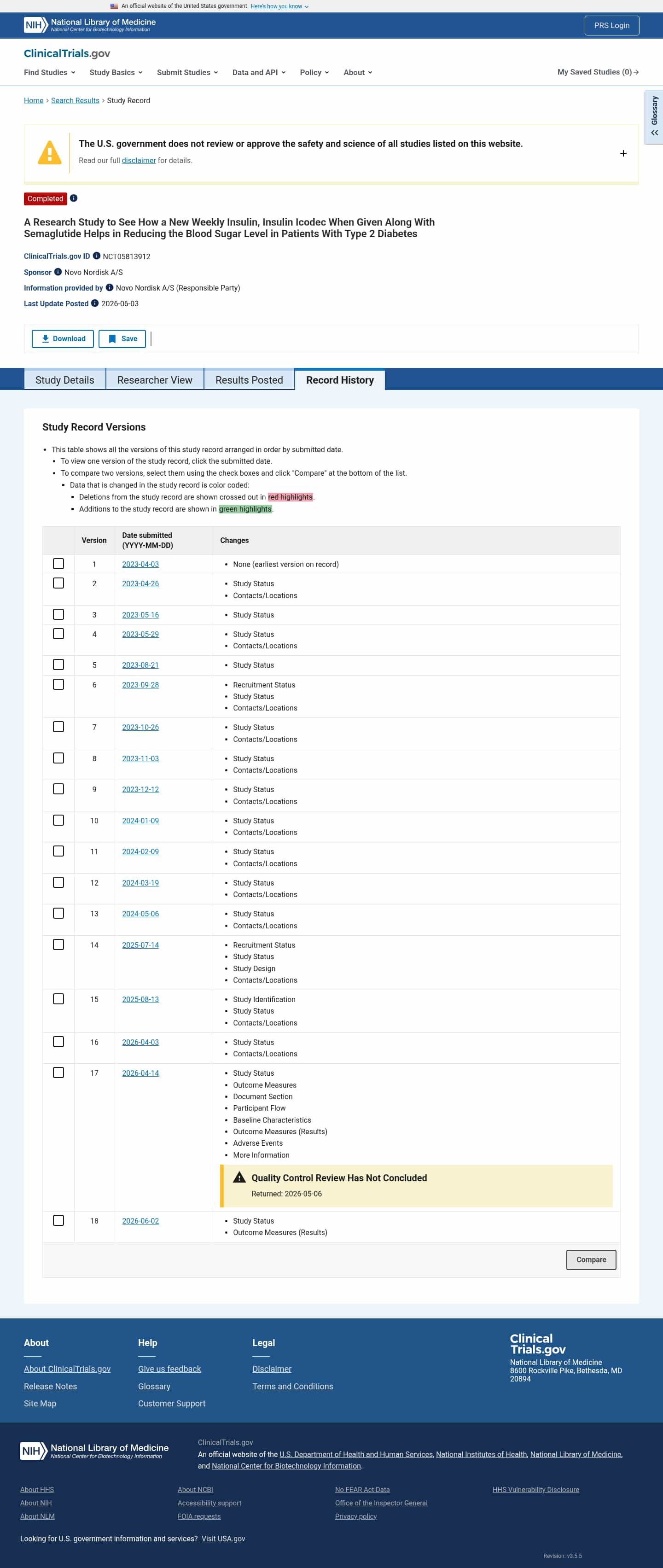
Latest updates to the Weekly Insulin Icodec with Semaglutide in Type 2 Diabetes Clinical Trial page
- Check2 days agoChange DetectedOn 2026-06-03, results have been posted and the Outcome Measures (Results) section is now populated. The record shows a Study Status update and a new Revision: v3.5.5.SummaryDifference0.6%
- Check9 days agoNo Change Detected
- Check16 days agoNo Change Detected
- Check23 days agoChange DetectedRevision badge updated from v3.5.3 to v3.5.4 at the bottom of the page. This reflects a new site version and does not change the page content or user interactions.SummaryDifference0.1%
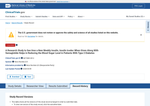
- Check31 days agoChange DetectedRecord History shows new entries and sections added to the study record, including Adverse Events, Outcome Measures (Results), Baseline Characteristics, Participant Flow, and More Information, plus a Quality Control Review Has Not Concluded status and a Returned date of 2026-05-06.SummaryDifference1%
- Check45 days agoChange DetectedThe ClinicalTrials.gov page footer has been updated to reflect a newer site/release version (v3.5.3 instead of v3.5.2). This represents a maintenance/release update rather than a change to the study record itself.SummaryDifference0.1%
- Check52 days agoChange DetectedAdded 'Results Submitted' and 'Revision: v3.5.2'; removed 'No Results Posted' and 'Revision: v3.5.0'.SummaryDifference0.2%
- Check59 days agoChange DetectedAdded and updated Study Status and Contacts/Locations entries in the record history (2026-04-03 and 2026-04-09), and removed an entry on 2025-08-17.SummaryDifference0.5%
Stay in the know with updates to Weekly Insulin Icodec with Semaglutide in Type 2 Diabetes Clinical Trial
Enter your email address, and we'll notify you when there's something new on the Weekly Insulin Icodec with Semaglutide in Type 2 Diabetes Clinical Trial page.