Best Sources for Clinical Trial News (2026)
By The Visualping Team
Updated June 19, 2023

A single clinical trial readout can move a biotech stock 40% in a session. A Phase 3 failure can wipe out billions in market cap before most investors even open their email. For researchers, a competitor's trial update might reshape your own study design. For patients, a new enrollment window could mean access to a treatment that did not exist last month.
Clinical trial news matters because the window between "published" and "priced in" keeps shrinking. The question is no longer whether you should track it, but how to track it without spending your entire day refreshing ClinicalTrials.gov.
This guide covers the best sources for clinical trial news, how to set up automated alerts for each one, and how to build a monitoring system that surfaces the updates you care about and filters out the rest.

Why staying current on clinical trial news matters
Researchers, investors, patients, and regulatory teams all track clinical trial news for different reasons, but they share one constraint: timing.
Researchers and pharma teams need trial updates to calibrate their own programs. When a competitor's Phase 2 trial reports positive interim data, it changes the competitive landscape for every similar molecule in development. Missing that update by a week could mean submitting a protocol amendment too late or pursuing an indication that just became crowded.
Investors and biotech analysts treat clinical trial results as primary catalysts. Binary events like PDUFA dates, data readouts, and FDA advisory committee meetings drive the majority of biotech stock volatility. Firms that monitor the original sources (SEC filings, company IR pages, FDA calendars) catch signals before the news wires pick them up.
Patients and caregivers track clinical trial news to find enrollment opportunities. Over 400,000 studies are registered on ClinicalTrials.gov as of 2026, and new trials open weekly. For patients with rare diseases or conditions where standard treatments have failed, a newly recruiting Phase 2 trial could represent their best option.
Regulatory and compliance teams use regulatory monitoring to stay ahead of guidance changes, safety signals, and evolving requirements from agencies like the FDA and EMA.
The challenge is the same for all four groups: clinical trial information is scattered across dozens of sources, published at unpredictable intervals, and buried in formats that range from structured databases to PDF press releases.
The 10 best sources for clinical trial news
Some sources publish structured data you can query. Others post updates as PDFs or press releases with no alerting mechanism. Here is where the most useful clinical trial information lives, and what each source is best for.
1. ClinicalTrials.gov
The U.S. National Library of Medicine's registry is the single largest source of clinical trial data globally. It contains over 400,000 registered studies, including trial status, enrollment criteria, study design, endpoints, and (increasingly) results summaries.
Best for: Tracking trial status changes (recruiting, active, completed, terminated), enrollment criteria updates, and posted results for specific trials or therapeutic areas.
Limitation: Status changes often lag behind real-world events. A trial might post interim results at a medical conference days before ClinicalTrials.gov reflects the update.
Alert mechanism: ClinicalTrials.gov offers RSS feeds for saved searches. You can create a search for a specific condition, sponsor, or intervention and subscribe to the feed for new or updated listings.
2. FDA press releases and safety alerts
The FDA's press announcements page covers approvals, Complete Response Letters (CRLs), safety communications, and advisory committee meeting outcomes. The FDA Drug Safety Communications page covers post-market safety signals.
Best for: Tracking approval decisions, breakthrough therapy designations, fast track grants, and safety actions that directly affect marketed drugs and late-stage trials.
Limitation: No native alerting beyond RSS. Press releases are published without a predictable schedule, so manual checking means missed updates.
3. PubMed and PubMed Central (PMC)
PubMed indexes over 37 million biomedical citations. Published trial results, systematic reviews, and meta-analyses appear here after peer review. PMC provides free full-text access to a growing subset.
Best for: Published clinical trial results, safety data, efficacy comparisons, and systematic reviews. PubMed is the gold standard for peer-reviewed evidence.
Alert mechanism: PubMed offers My NCBI email alerts that notify you when new articles match a saved search query.
4. SEC EDGAR (investor-relevant filings)
For publicly traded biotech and pharma companies, the SEC's EDGAR database contains 8-K filings, 10-K/10-Q reports, and prospectus supplements that often disclose clinical trial results, regulatory interactions, and pipeline updates.
Best for: Investors and analysts tracking material clinical events for publicly traded companies. An 8-K filing with trial data often precedes the press wire by minutes.
Alert mechanism: EDGAR offers company-specific RSS feeds for new filings. For structured alerts, see the SEC Form 4 alert workflow that applies to any EDGAR page.
5. Clinical Trials Arena
Clinical Trials Arena is a dedicated news site covering trial design, regulatory developments, technology, and therapeutic area updates. It publishes original reporting alongside data-driven analysis.
Best for: Industry professionals who want curated, context-rich clinical trial news rather than raw data feeds.
6. STAT News
STAT News covers health, medicine, and the life sciences with a strong focus on drug development and clinical research. Their "First Opinion" and investigative pieces often break stories about trial safety concerns or regulatory decisions.
Best for: Breaking clinical trial news, FDA policy analysis, and investigative coverage of trial conduct issues.
7. BioPharma Dive
BioPharma Dive covers the pharmaceutical and biotech industries, including a dedicated clinical trials topic page with daily updates on trial results, pipeline moves, and regulatory filings.
Best for: Quick daily briefings on the most important clinical trial developments across the industry.
8. Nature and The New England Journal of Medicine (NEJM)
Nature and NEJM publish the highest-impact clinical trial results. Landmark trials (KEYNOTE, RECOVERY, SPRINT) appear here first. These publications carry significant weight with regulators, payers, and the medical community.
Best for: Definitive clinical trial results that shape treatment guidelines and standard of care.
9. Company IR pages and press rooms
Publicly traded pharma and biotech companies post clinical trial results, pipeline updates, and regulatory milestones on their Investor Relations pages, often before (or simultaneously with) wire services. For companies in your coverage universe, these pages are primary sources.
Best for: Company-specific trial updates, conference presentation slides, earnings call transcripts mentioning pipeline data, and real-time PR distributions.
Most IR pages have no alert mechanism. Unless you check manually or use a website monitoring tool, you will miss updates between scheduled checks.
10. WHO International Clinical Trials Registry Platform (ICTRP)
The WHO ICTRP aggregates trial registrations from national registries worldwide, including the EU Clinical Trials Register, Japan's JPRN, and China's ChiCTR. It provides a global view beyond ClinicalTrials.gov's primarily U.S.-focused database.
Best for: Tracking international clinical trials, especially for global pharma companies running multi-region studies or researchers monitoring trial activity in specific countries.

How to set up automated clinical trial alerts
Knowing where to look is only half the problem. The other half is getting notified when something changes without manually checking ten different sources every morning. Here are four approaches, from basic to fully automated.
ClinicalTrials.gov RSS feeds
Create a saved search on ClinicalTrials.gov filtered by condition, sponsor, intervention, or status. Click "Create alert" to generate an RSS feed. Add this feed to an RSS reader (Feedly, Inoreader) or connect it to Slack via an RSS integration.
Covers: New trial registrations, status changes, and results postings for your saved search criteria.
Misses: Changes to existing trial pages that do not trigger the RSS feed (enrollment criteria updates, site additions, protocol amendments).
PubMed email alerts
Register for a free My NCBI account and save your clinical trial search queries. Configure email alerts for daily or weekly delivery when new publications match your query.
Covers: Published results, systematic reviews, and safety reports.
Misses: Preprints, conference abstracts, and results posted to registries before formal publication.
Google Alerts
Set up a Google Alert for terms like "Phase 3 results [drug name]" or "clinical trial [company name]." Google will email you when new web pages matching your query appear in its index.
Covers: News articles, press releases, and blog posts mentioning your terms.
Misses: Changes to existing pages (updated trial statuses, revised enrollment criteria). Google Alerts only catches new URLs, not modifications to pages you already know about.
Monitoring clinical trial pages with Visualping
The gap in every alerting method above is the same: they catch new content but miss changes to existing pages. When ClinicalTrials.gov updates a trial's status from "Recruiting" to "Active, not recruiting," RSS may not fire. When an IR page swaps out a slide deck with updated interim data, Google Alerts won't notice.
Visualping fills this gap. It monitors any web page at intervals you choose (from every 5 minutes to daily) and sends you an alert when the content changes. The alert includes a screenshot with changes highlighted and an AI-generated summary explaining what changed and why it matters.
How to set it up:
- Copy the URL of the page you want to monitor (a ClinicalTrials.gov study page, an FDA announcement page, a company's IR news section)
- Paste it into Visualping and select the specific section of the page to watch
- Tell the AI what changes matter to you (for example: "Alert me when the trial status changes" or "Alert me when a new press release about Phase 3 data appears")
- Choose your check frequency and alert channel (email, Slack, webhook)
Visualping works on any public web page, so it covers sources that have no native alert mechanism: company IR pages, FDA guidance documents, regulatory agency calendars, conference presentation archives, and niche databases.
Building a clinical trial monitoring dashboard
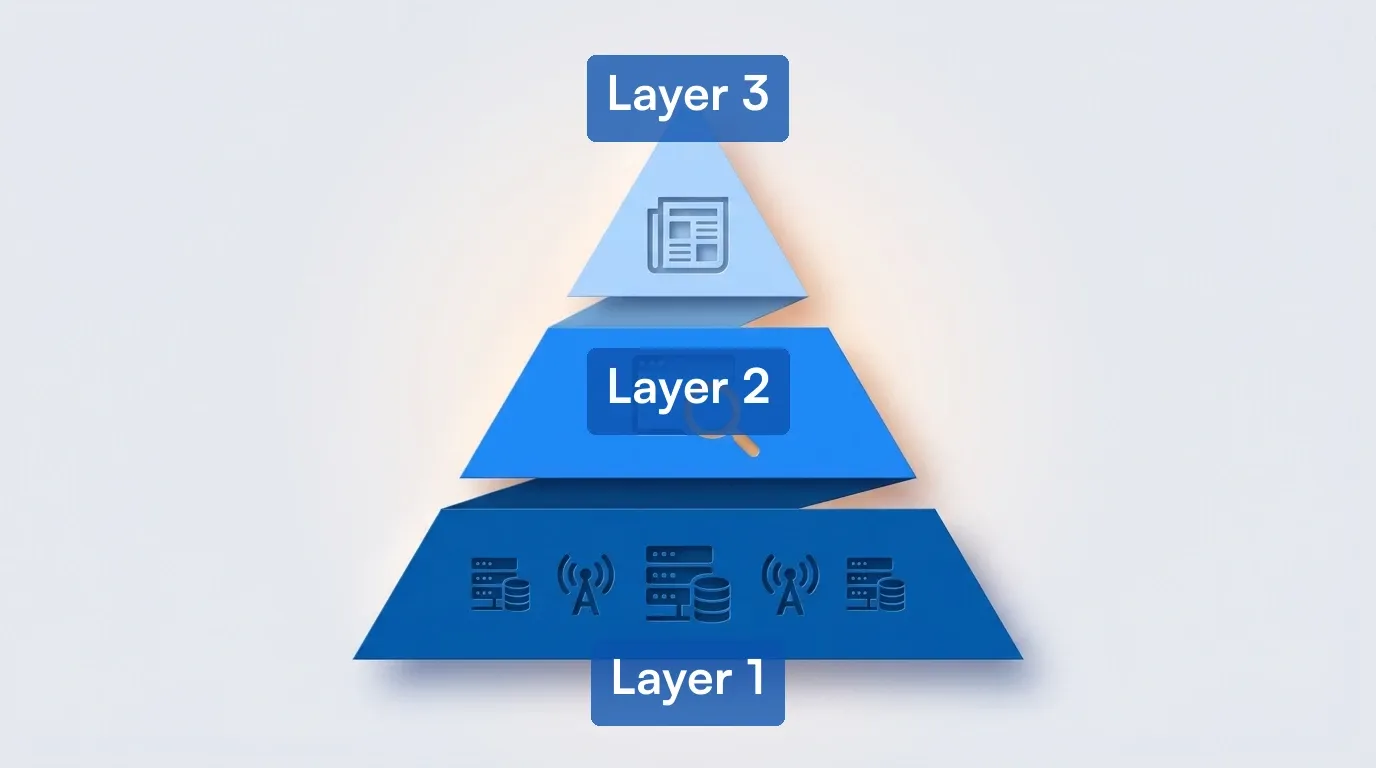
Tracking ten sources individually gets messy fast. Here is a three-layer framework that pulls them into one workflow.
Layer 1: Structured feeds (automatic)
- ClinicalTrials.gov RSS feeds for your therapeutic areas and target companies
- PubMed email alerts for published results in your focus area
- SEC EDGAR RSS for pipeline-relevant 8-K and 10-K filings
Layer 2: Web monitoring (automated via Visualping)
- Company IR pages for your coverage universe (5-minute to hourly checks)
- FDA press announcements and advisory committee calendars (hourly checks)
- ClinicalTrials.gov individual study pages (daily checks for status changes)
- Conference presentation archives during major medical meetings (ASCO, ASH, ESMO, AHA)
Layer 3: Curated news (daily review)
- Clinical Trials Arena, STAT News, BioPharma Dive daily briefings
- Journal table of contents alerts from NEJM, The Lancet, Nature Medicine
Routing alerts to your workflow: Visualping integrates with Slack, Google Sheets, Zapier, and n8n. You can route clinical trial alerts into a dedicated Slack channel, log them to a spreadsheet for historical tracking, or trigger downstream workflows (like updating an investment model or notifying a research team).
For teams running event-driven investing strategies, this three-layer setup covers databases, primary sources, and news outlets in a single workflow.
Who tracks clinical trial news (and why)
Biotech investors tracking Phase 2/3 readouts
A biotech-focused hedge fund monitors 30 companies across oncology and rare disease. For each company, they watch the IR page, the relevant ClinicalTrials.gov study page, and SEC EDGAR for material filings. Visualping checks these pages every 15 minutes during market hours and routes alerts to a shared Slack channel. When a company posts interim Phase 2 data to its IR page at 6:45 AM, the team gets the alert at 6:47 AM, two full hours before the press release hits the newswire at 9:00 AM.
Pharma R&D teams watching competitor pipelines
A pharmaceutical company's competitive intelligence team tracks 12 competitor programs in the same therapeutic area. They monitor ClinicalTrials.gov for status changes, PubMed for published results, and competitor IR pages for conference presentations. When a competitor's trial moves from Phase 2 to Phase 3, the team adjusts their own development timeline and resource allocation within hours.
Patient advocacy groups monitoring trial access
A rare disease patient foundation monitors ClinicalTrials.gov for new trials matching their condition. When a new study begins recruiting in their geography, they alert their member community within the day. They also watch FDA pages for Expanded Access and Compassionate Use announcements that could give patients access to experimental treatments outside of formal trials.
Regulatory teams tracking FDA guidance changes
A compliance team at a mid-size pharma company monitors FDA guidance documents, advisory committee schedules, and Federal Register notices. When the FDA publishes a draft guidance that affects their submission strategy, they catch it the same day rather than learning about it from a trade publication the following week.
Go deeper: What Is Regulatory Intelligence and Compliance? | Build Custom Data Feeds for Investing
Frequently asked questions
Where is the best place to find clinical trial news?
ClinicalTrials.gov is the largest source for trial registrations and status updates. For breaking news and analysis, STAT News and Clinical Trials Arena provide daily coverage. For published results, PubMed and major journals (NEJM, The Lancet) are the standard. For investor-relevant filings, SEC EDGAR captures material disclosures from publicly traded companies.
How do I get alerts when a clinical trial status changes?
ClinicalTrials.gov offers RSS feeds for saved searches, which catch new registrations and some status changes. For page-level monitoring that catches all modifications (including status updates, enrollment criteria changes, and results postings), use a website change monitoring tool to watch the specific study page.
Can I track clinical trials for a specific drug or condition?
Yes. On ClinicalTrials.gov, search by condition, intervention, or sponsor name and save the search. On PubMed, create a saved search with your terms. For company-specific updates, monitor the company's IR page and SEC filings. Tools like Visualping let you set keyword filters so you only get alerted when changes match your specific criteria.
How often are clinical trial databases updated?
ClinicalTrials.gov requires sponsors to update records within 30 days of a status change, though compliance varies. FDA press announcements publish in real-time when decisions are made. PubMed indexes new publications daily. Company IR pages update unpredictably, which is why automated monitoring is valuable for catching updates the moment they appear.
What is the difference between ClinicalTrials.gov and the WHO ICTRP?
ClinicalTrials.gov is the U.S. registry maintained by the National Library of Medicine. The WHO ICTRP aggregates registrations from 17 national and regional registries worldwide, giving a global view. If you are tracking international trials (especially in the EU, Japan, or China), the WHO ICTRP covers registries that ClinicalTrials.gov may not include.
How can investors use clinical trial news for investment decisions?
Investors track clinical trial catalysts (Phase 2/3 data readouts, PDUFA dates, advisory committee meetings) as binary events that drive biotech stock prices. Monitoring original sources like SEC EDGAR, company IR pages, and ClinicalTrials.gov directly provides earlier access to information than waiting for news wire coverage. For a detailed workflow, see our guide on event-driven investing with web data.
Conclusion
Clinical trial news is scattered across registries, regulatory agencies, journals, news sites, and corporate IR pages. No single source covers everything, and most sources have no alerting mechanism beyond basic RSS feeds.
The practical solution is a layered monitoring system: structured feeds for databases that support them, automated web monitoring for pages that do not, and curated news for context and analysis. Tools like Visualping close the gap between sources that offer alerts and those that do not, ensuring you catch updates from any web page the moment they change.
Set up the three layers this week. Start with a ClinicalTrials.gov RSS feed for your therapeutic area, add Visualping on two or three IR pages you currently check by hand, and subscribe to one daily briefing. The whole system takes about 30 minutes to build and replaces hours of manual refreshing.
Want to monitor web changes that impact your business?
Sign up with Visualping to get alerted of important updates, from anywhere online.
The Visualping Team
Visualping helps over 2 million users monitor websites for changes. Our platform powers competitive intelligence, compliance monitoring, and automated research workflows for teams across finance, pharma, and technology.