What Is Compliance Monitoring? A Complete Guide - Visualping
By The Visualping Team
Updated May 17, 2024

A single missed regulatory update can cost your organization six figures in fines, or worse. Every business operates under federal, state, and industry regulations that shift constantly. Compliance monitoring is the practice of systematically tracking those updates, verifying internal policy adherence, and catching enforcement actions before they catch you.
Compliance monitoring is the ongoing process of reviewing and verifying that a business meets all applicable laws, regulations, and internal policies. It involves tracking regulatory changes, auditing internal practices, and documenting activities to reduce risk and avoid violations.
How this guide was prepared. Penalty figures, enforcement statistics, and program standards cited here come from primary sources: the U.S. Securities and Exchange Commission, the Occupational Safety and Health Administration, the Department of Health and Human Services Office of Inspector General, the UK Financial Conduct Authority, and the U.S. Environmental Protection Agency. Operational benchmarks come from anonymized Visualping platform data covering 1.8 million active monitors as of March 2026. Last reviewed May 2026.
This guide walks through the full program: what to track, how often to check, and a seven-step framework you can adapt to your team. (It covers general compliance monitoring practices. Consult your legal or compliance team for advice specific to your regulatory obligations.)
How big does a compliance monitoring program need to be? Visualping platform data from April 2026 spans 1.8 million active monitors. Among business accounts tracking regulatory pages, 22% watch a single critical source, 23% monitor 6 to 20, and 14% track over 100. Most teams start small and bolt on more sources as they identify gaps.

Why Compliance Monitoring Matters
Non-compliance carries real consequences. The U.S. Securities and Exchange Commission (SEC) obtained over $8.2 billion in financial remedies during fiscal year 2024, a record high. OSHA can fine employers up to $165,514 per willful violation. These are not theoretical risks.
Beyond enforcement, a monitoring program protects organizations in ways that are harder to measure but just as real.
It cuts financial exposure. Catching a regulatory change before it takes effect gives your team time to adjust policies, retrain staff, and update procedures. Organizations that learn about changes after enforcement begins pay both the penalty and the cost of rushed remediation.
It builds external trust. Customers, partners, and investors look at how well a company manages regulatory risk. A documented program shows that your house is in order, particularly in industries like regulatory compliance in banking and pharmaceutical compliance where regulatory expectations are public.
It also catches internal policy drift: day-to-day practices that gradually wander from documented procedures. Regular reviews surface these gaps before they become audit findings.
The regulatory environment is also getting noisier. In October 2025, 2,409 Visualping compliance monitors detected at least one change on a government or regulatory page. By March 2026, that number reached 39,307, a 16-fold increase in six months (Visualping platform data). Manual tracking habits that worked a few years ago cannot absorb that volume.
Over 10,600 Visualping users selected "Laws & Regulations" as their primary reason for monitoring during onboarding (Visualping onboarding data, March 2026).
The 7 Elements of an Effective Compliance Program
Compliance monitoring sits inside a broader program. The U.S. Department of Health and Human Services Office of Inspector General published seven elements that have become the de facto standard for compliance programs across industries, not just healthcare. They are:
- Written standards and procedures. Policies that document what compliance looks like for your organization, written in language your team can actually apply.
- Compliance leadership and oversight. A designated compliance officer or committee with authority to act, plus board-level oversight for material risks.
- Training and education. Role-specific training so each function understands the regulations that apply to their work, refreshed when rules change.
- Open lines of communication. A reporting channel for employees to raise concerns without retaliation, plus a clear escalation path to leadership.
- Monitoring and auditing. The continuous and point-in-time review activities this guide focuses on. Monitoring is the daily heartbeat; auditing is the periodic deep dive.
- Consistent enforcement and discipline. Defined consequences for violations, applied uniformly. A program that disciplines selectively erodes faster than one with no rules at all.
- Prompt response and corrective action. When issues surface, a documented remediation process, including root-cause analysis and policy updates, closes the loop.
Monitoring (element 5) only works if the other six are in place. A monitoring program without named owners (element 2), trained staff (element 3), or response procedures (element 7) generates alerts that no one acts on.
Types of Compliance Monitoring
The approach depends on what you are tracking, who owns it, and how frequently changes land.
External Compliance Tracking
The most common form. Your team watches for updates to laws, regulations, and enforcement guidance from government agencies: the Federal Register for proposed rulemakings, FDA guidance documents, state legislature websites for new bills.
Regulatory pages change more often than most teams realize. Across the Visualping platform, 39.3% of government and regulatory pages detected at least one change in the past 30 days (Visualping platform data, March 2026). That is roughly two out of every five monitored pages updating in a single month.
One thing worth knowing: web pages and PDF documents behave very differently. Among Visualping regulatory monitors in the 30 days ending April 2026, web pages changed 41% of the time. PDFs changed just 10.4%. PDFs change 4x less often, but when a regulatory PDF does update, it is almost always substantive: a new filing, an amended regulation, or revised guidance. Set lower check frequencies for PDFs, but treat every change as high-priority.

Internal Policy Adherence
Here the focus turns inward: are employees and departments actually following the company's own policies? Internal reviews typically involve checking training completion records, auditing expense reports against spending policies, and verifying that standard operating procedures match what people actually do.
Third-Party and Vendor Oversight
Your vendors, suppliers, and partners carry regulatory obligations too, and their failures become your problem. Vendor oversight matters most in financial services (Know Your Customer requirements), healthcare (HIPAA Business Associate Agreements), and technology (data processing agreements under GDPR).
License and Certification Tracking
Software licenses, professional certifications, and industry accreditations all have renewal dates and changing terms. Missing a renewal or operating under expired terms creates gaps that auditors will flag. You can track these manually or with AI web monitoring tools that alert you when terms shift.
How to Build a Compliance Monitoring Plan
Building an effective plan does not require a massive budget or a dedicated department. This seven-step framework scales with your organization. Adapt the depth of each step to match your team size and regulatory complexity.
Step 1: Identify Your Regulatory Universe
Start by cataloging every regulation, law, and standard that applies to your business. Group them by source: federal, state, local, and international. Include industry-specific regulations (SOX for public companies, HIPAA for healthcare, PCI-DSS for payment processing) and broadly applicable ones like:
- Anti-discrimination policies from the Equal Employment Opportunity Commission (EEOC)
- Wage requirements under the Fair Labor Standards Act (FLSA)
- Workplace safety rules defined by the Occupational Safety and Health Administration (OSHA)
This catalog becomes the foundation of your monitoring plan. If you do not know what applies to you, you cannot track it.
Step 2: Assess and Prioritize Risk
Not every regulation carries the same risk. A missed OSHA reporting deadline has different consequences than a late filing with the SEC. Score each regulatory area on two axes: how likely a change is and how severe non-compliance would be.
The risk profile looks different by industry. Banking compliance centers on capital adequacy and anti-money laundering. Pharmaceutical regulatory intelligence revolves around drug approval timelines and clinical trial requirements. Prioritize the areas where a violation would hit hardest.
Step 3: Assign Ownership
Every regulatory area needs a named owner: someone responsible for tracking changes, assessing impact, and escalating when action is needed. In larger organizations, this maps to a compliance officer or a cross-functional committee. In smaller companies, it might fall to legal counsel, an operations lead, or the founder.
Clear ownership prevents the "I thought someone else was watching that" problem. From what we have seen working with compliance teams, this single gap causes more regulatory misses than any other.
Step 4: Set Monitoring Frequency
How often you check should match how quickly regulations move in your industry. Our data shows that most monitors for government pages check every 1 to 24 hours (60.7%), while over a third (37.2%) check sub-hourly (Visualping platform data, March 2026). That sub-hourly group tells you something about the stakes some teams are dealing with.
To help calibrate, here is how often major regulatory domains actually change, based on monitors running on the Visualping platform (Visualping platform data, March 2026):
| Regulatory Source | Active Monitors | Changed in Last 30 Days | Median Check Frequency |
|---|---|---|---|
| Federal Register | 154 | 61.7% | Every 24 min |
| canada.ca | 3,567 | 60.6% | Every 2.8 hrs |
| FTC | 257 | 50.2% | Every 24 min |
| europa.eu | 5,450 | 43.5% | Every 24 min |
| SEC | 1,003 | 42.9% | Every 2.8 hrs |
| FDA | 3,007 | 36.1% | Every 24 min |
| EPA | 619 | 30.9% | Every 2.8 hrs |
| IRS | 1,441 | 27.3% | Every 24 min |
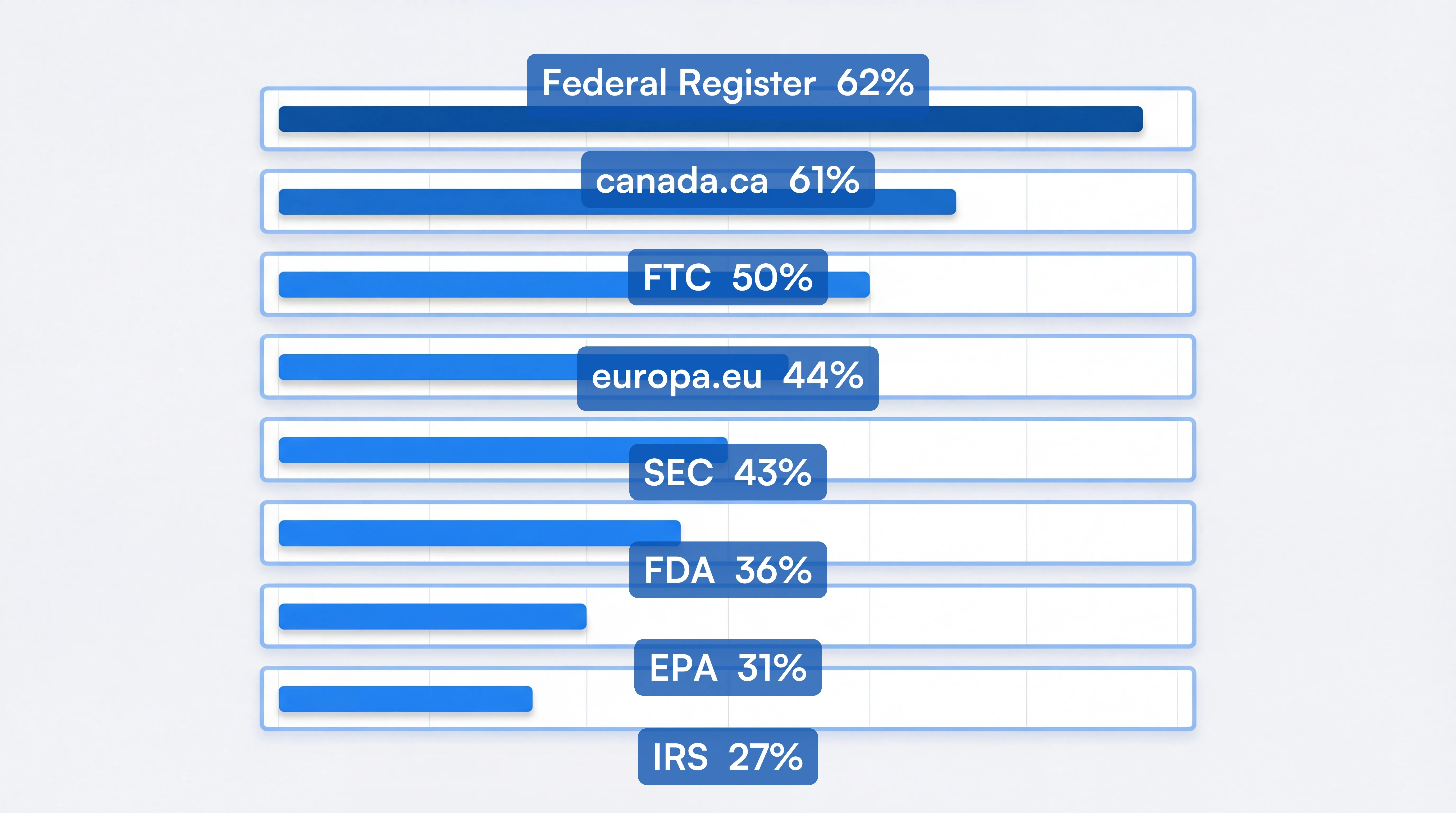
The Federal Register and FTC change the most frequently. FDA and SEC are mid-frequency but high-impact. Use these benchmarks as a starting point:
- High-risk, fast-changing (Federal Register, FTC, SEC): Check every 1 to 6 hours
- Medium-risk, periodic (FDA, EPA, state legislature sessions): Check daily
- Low-risk, stable (professional licensing terms, industry standards): Check weekly
Step 5: Automate Regulatory Change Detection
Manually checking government websites and agency announcements does not scale when your team tracks dozens or hundreds of regulatory sources. Automation handles the detection: watching the pages, flagging changes, logging timestamps. That frees the compliance team to spend their hours on interpretation and response.
Visualping's regulatory intelligence platform checks regulatory web pages at the frequency you set and sends alerts when content changes. Over 171,000 active monitors on the platform track government and regulatory pages, and two-thirds (66.2%) of those belong to business teams rather than individual users (Visualping platform data, March 2026). At that scale, compliance monitoring is an organizational capability, not a personal habit.
For teams that need to wire up monitors across dozens or hundreds of regulatory sources, the Visualping API makes this practical. Instead of clicking through a dashboard to create each monitor one by one, a single script can onboard your entire regulatory universe in one run:
import requests
API_KEY = "YOUR_API_KEY"
headers = {
"Authorization": f"Bearer {API_KEY}",
"Content-Type": "application/json"
}
regulatory_sources = [
{"url": "https://www.sec.gov/rules", "name": "SEC rulemaking"},
{"url": "https://www.federalregister.gov/", "name": "Federal Register"},
{"url": "https://www.fda.gov/regulatory-information", "name": "FDA guidance"},
]
for source in regulatory_sources:
payload = {
"url": source["url"],
"description": source["name"],
"interval": "360",
"mode": "ALL",
"target_device": "4",
"wait_time": 0,
"summalyzer": {
"importantDefinitionType": "custom",
"importantDefinition": "New rules, amended regulations, enforcement actions, or policy changes"
},
"notification": {
"onlyImportantAlerts": True
}
}
response = requests.post(
"https://job.api.visualping.io/v2/jobs",
headers=headers,
json=payload
)
if response.status_code == 200:
print(f"Monitor created: {source['name']}")
The importantDefinition field tells the AI what counts as a material regulatory change. Formatting tweaks and footer updates stay silent. Only substantive changes (new rules, amended language, enforcement actions) fire alerts. The API also supports webhook routing, bulk creation, and AI-generated change summaries.
Among Visualping compliance monitors (April 2026), 16.7% use the AI summarizer to classify changes by importance. 12.8% filter for only the changes flagged as material. For teams watching dozens of regulatory sources, that filtering cuts alert fatigue without missing substantive updates.
Step 6: Create Reporting and Documentation Protocols
Reporting does two things: it proves monitoring is happening, and it creates an audit trail for regulators and internal teams.
A good report includes the issue identified, the action taken, the resolution, and the timeline. Your reporting protocol should specify who compiles reports, how they are stored, and who has access.
Documentation matters most when regulators come knocking. "We have a monitoring program" (with timestamped logs to prove it) carries far more weight with auditors than "we try to stay informed."
Step 7: Measure and Improve
Track key performance indicators (KPIs) to see whether your program is actually working. Useful KPIs include:
- Time to detect a regulatory change (detection lag)
- Time to assess impact and implement changes (response time)
- Number of violations or near-misses per quarter
- Employee complaints and their resolution rates
- Audit findings trend (improving or worsening)
Review these metrics quarterly. A compliance monitoring plan is not a one-time project. Regulations change, your business evolves, and your program has to keep pace.
Government and Regulator-Led Monitoring
Compliance monitoring also runs in the other direction. Federal and state agencies operate their own monitoring programs to verify that regulated entities follow the rules. The U.S. Environmental Protection Agency, for example, runs on-site inspections, off-site data reviews, and self-disclosure incentive programs. State agencies do the same for environmental, financial, healthcare, and labor regulations.
Two implications for businesses:
- You are being monitored. Most regulators publish inspection schedules, enforcement actions, and audit results on their websites. Tracking those pages (for your own facilities and your industry peers) gives early warning of where regulators are focusing attention.
- Your monitoring is being reviewed. When regulators audit you, they look for documented evidence that your internal monitoring program exists and operates. The artifacts your program produces (review logs, change records, response documentation) are what auditors evaluate.
Manual vs. Automated Approaches
One of the first decisions in building a program: manual processes or automation? Here is how they compare:
| Factor | Manual Approach | Automated Approach |
|---|---|---|
| Coverage | Limited to what staff can review in available hours | Can track hundreds or thousands of regulatory sources simultaneously |
| Speed | Hours to days between a change and discovery | Minutes to hours, depending on check frequency |
| Cost | Staff time (often expensive at senior compliance rates) | Platform subscription plus setup time |
| Accuracy | Prone to human oversight, especially for subtle changes | Detects any change, including formatting or embedded content shifts |
| Scalability | Adding sources means adding headcount | Adding sources requires minimal incremental effort |
| Audit trail | Requires manual logging of every check performed | Automatic timestamped logs of every check and change detected |
| Programmatic setup | Not applicable | API-driven bulk creation for 50+ sources |
Most organizations use a combination. Automated tools handle the detection layer (watching for changes); compliance teams handle the interpretation layer (deciding what a change means and how to respond). That split puts judgment where it matters most. For a deeper look at available platforms, see our guide to compliance monitoring tools and software.
Visualping's AI classifies over 242,000 active monitors as tracking government, legal, or procurement content (Visualping platform data, March 2026).

Compliance Monitoring Best Practices
Beyond the seven-step framework, these practices tighten any compliance monitoring effort.
Keep your regulatory catalog current. New regulations appear, old ones get amended, and some get repealed entirely. Schedule a quarterly review of your regulatory universe to make sure nothing has fallen off the radar. Horizon scanning helps teams spot upcoming changes before they take effect.
Centralize your monitoring sources. Scattered tracking across email subscriptions, bookmarked websites, and individual staff knowledge creates blind spots. One platform or dashboard where all regulatory sources are visible to the team kills the "I didn't know we were supposed to watch that" problem.
Train beyond the dedicated team. Front-line employees are often the first to encounter compliance issues. Regular training ensures they recognize problems and know how to escalate. Track training completion as a KPI.
Document everything, even when nothing changes. A check that finds no updates is still valuable evidence for auditors. Automated tools create this documentation by default, logging every check whether or not a change was detected.
Coordinate with related functions. Compliance monitoring overlaps with financial compliance, risk management, internal audit, and legal. Regular coordination prevents duplicated effort and plugs the gaps between functions.

Common Compliance Monitoring Challenges
Even well-designed programs run into recurring problems. The pattern repeats across industries:
- Information overload. Tracking dozens of regulatory sources generates a high volume of alerts, and not every change is material. Effective programs filter aggressively: separating substantive amendments from typo fixes and announcement noise.
- Resource constraints. Compliance monitoring takes financial, human, and technical investment. Smaller teams struggle to allocate dedicated headcount, which is why automation has become the default starting point for organizations without a full compliance department.
- Regulatory complexity across jurisdictions. Multinational operations sit under overlapping rules (SEC plus FCA plus ESMA, for example). Each jurisdiction publishes on its own cadence and in its own format, which makes a single source-of-truth dashboard hard to build manually.
- Manual processes that don't scale. Spreadsheet-based tracking introduces errors, missed updates, and stale documentation. The cost of a missed amendment is rarely the spreadsheet; it's the downstream enforcement action.
- Accountability gaps. Monitoring requires named owners. When responsibility is diffuse, regulatory changes get noticed but not actioned. Programs that work assign each regulatory source to a specific person with a specific review cadence.
- Integration with existing systems. Compliance data lives in GRC platforms, ticketing systems, document repositories, and email. Pulling these together into a single audit trail is the most common technical bottleneck reported by compliance teams.
Information overload is the most common. Tracking dozens of regulatory sources generates a high volume of alerts, and not every change is relevant to your organization. Effective programs require filtering: separating material changes from minor formatting updates or procedural corrections. Tools with visual change comparison and keyword-based alerts help cut through the noise.
Resource constraints hit smaller teams hardest. Companies without dedicated departments struggle to carve out time for this work. Automation handles the detection problem, but someone still needs to interpret changes and act on them. Consider bringing in external consultants for specialized regulatory areas where in-house expertise is thin.
Cross-jurisdictional complexity catches growing organizations off guard. Operating in multiple states or countries means overlapping and sometimes conflicting regulations, as real-world examples of regulatory compliance across industries illustrate. A California data privacy update may conflict with how your team handles GDPR in Europe. Build your plan with jurisdictional layers so each regulatory area has clear scope and ownership.
Regulatory velocity is harder to predict than most teams expect. Maintaining regulatory compliance is an ongoing challenge because agencies issue updates at irregular intervals. Some publish frequently (the SEC filed 583 total enforcement actions in fiscal year 2024), while others update rarely but with major consequences. Your check frequency should reflect each source's actual publication cadence.
Frequently Asked Questions
What is the difference between compliance monitoring and a compliance audit?
Monitoring is continuous. It involves ongoing tracking of regulatory changes and internal policy adherence, typically through automated tools and regular reviews. An audit is a point-in-time snapshot: a structured evaluation of whether an organization meets specific requirements as of a particular date. Monitoring feeds into audits by providing the ongoing data that auditors review.
How often should a company review its program?
Most professionals recommend a formal review at least quarterly, with an annual review that reassesses the full regulatory universe. Trigger-based reviews matter too: any time your business enters a new market, launches a new product, or faces a regulatory enforcement action, revisit the plan.
What industries require this type of monitoring?
Every industry has some regulatory requirements, but structured programs are most critical in financial services, healthcare, pharmaceuticals, energy, manufacturing, and any sector handling personal data. See our overview of website change detection for how automated monitoring fits into these industries. The SEC, FDA, EPA, and OSHA are the regulatory bodies companies track most often. Life sciences runs deep on Visualping: clinicaltrials.gov has over 7,500 active monitors, cms.gov (Medicare and Medicaid) over 3,000, and ema.europa.eu (European Medicines Agency) nearly 1,000 (March 2026).
Can small businesses benefit from compliance monitoring?
Yes. Small businesses face the same regulations as larger companies but have fewer people to track changes. Automated tools close that gap by trimming the manual effort. A small business can set up monitors on the specific regulatory pages relevant to their industry and get alerts when anything changes, no dedicated department required.
What should a monitoring report include?
A report should include:
- The regulatory source being tracked
- The date and nature of any changes detected
- An impact assessment (does this change affect our operations?)
- The action taken in response
- The responsible party
- The completion date For periods with no changes, the report should still document that the review occurred. That "nothing changed" log is exactly what auditors want to see.
Get ahead of the next regulatory change
Building a compliance monitoring program takes planning. Discovering regulatory changes after a violation takes lawyers.
Visualping lets you monitor regulatory changes from any government or regulatory website. Set your check frequency, add the pages that matter to your industry, and get alerts when anything changes. Over 171,000 regulatory monitors run on the platform today. Teams managing larger regulatory portfolios use the API to create monitors programmatically and route alerts to Slack, Teams, or any webhook endpoint.
For a look at tools purpose-built for this workflow, see our guide to compliance monitoring software solutions.
Want to monitor web changes that impact your business?
Sign up with Visualping to get alerted of important updates from anywhere online.
The Visualping Team
Trusted by 2M+ users for competitive intelligence, compliance monitoring, and automated workflows.